Links:
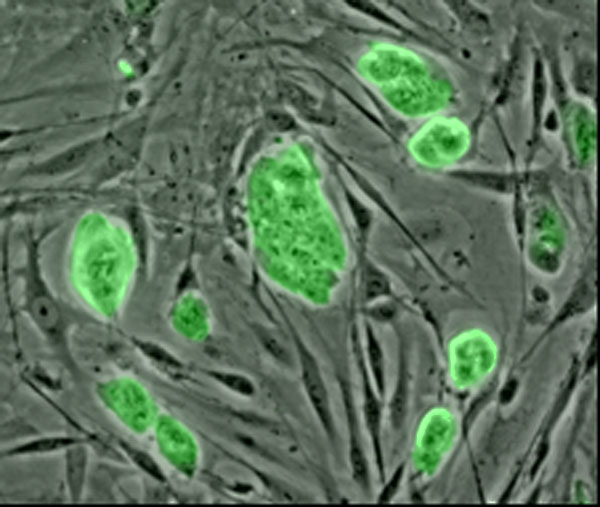
The ability to renew themselves through mitotic cell division and can differentiate into a diverse range of specialized cell types
Source:
Source: |
New Scientist video round-up - November 16, 2007
Which primate has the fastest sperm?
ICSI (Sperm Injection) for
Male Infertility
Intracytoplasmic sperm injection (ICSI)
Stem Cell Injection in China
Steam Cell Reasearch
The ability to create living tissues and organs in the lab

Photo courtesy of technologyreview.com

Photo courtesy of Mehrnews

Photo courtesy of Mehrnews
This animation of a rotating Carbon nanotube shows
its 3D structure, Courtesy of Wikipedia
The ability to create living tissues and organs in the lab holds great promise for transplant medicine. But the traditional approach to tissue engineering--seeding the outside of a biodegradable scaffold with cells, without regard to their organization--hasn't gotten cells to behave the way they would in the body. |

Courtesy of technologyreview.com
Ali Khademhosseini, assistant professor in medicine and health sciences and technology at Harvard Medical School, hopes to improve engineered tissues with an approach he likens to building with living Legos. As a first step toward creating a heart, he aligns cardiac muscle cells to form small, beating strings. He then embeds these strings in a supportive, gelatinous polymer to make building blocks that can be assembled into bundles resembling the sheets of muscle that make up the heart. He can also add other types of cells to the building blocks to provide support for the muscle. This aspect of the system is crucial, since natural tissues comprise cells of multiple types in structurally complex arrangements. By giving cells the same interconnections they have in the body, Khademhosseini hopes to create tissues that can be used to test new drugs and, eventually, to rebuild organs. Source: |
Functionalization of the biomaterials for tissue engineering scaffolds, Prof. Ali Khademhosseini, Harvard University, USA. His research: The development of micro- and nanoscale technologies to control cellular behavior with particular emphasis in developing microscale biomaterials and engineering systems for tissue engineering and drug delivery
Micro- and Nanoscale Technologies for 3D Tissue Engineering
Ali Khademhosseini, Ph.D., Assistant Professor, Harvard / MIT Division of Health Sciences and Technology, Brigham and Women’s Hospital
Tissues are highly organized in their geometry and architecture with respect to how cells are positioned relative to each other, as well as to the surrounding soluble factors and extracellular matrix molecules within a given microenvironment. Most existing methods of generating tissues in 3D have not been able to recapitulate the proper microstructure and function of 3D tissues in the body. The merger of microscale technologies and novel biomaterials is a potential approach to generate tissues that mimic the complexity of tissues in the body. The talk will describe the current state-of-the-art in the application of microscale technologies for 3D cell culture. Specifically, I will describe our work in controlling the 3D cellular microenvironment by encapsulation within engineered microscale biomaterials, by using microstructures to generate homogeneous microtissues, by controlling the spatial distribution of cells and molecules within hydrogels and by directly engineering the microvasculature into 3D structures.
Source:
Ali Khademhosseini, Ph.D
Ali Khademhosseini is an Assistant Professor of Medicine and Health Sciences and Technology at Harvard-MIT's Division of Health Sciences and Technology and the Harvard Medical School. His research area includes the development of micro- and nanoscale technologies to control cellular behavior with particular emphasis in developing microscale biomaterials and engineering systems for tissue engineering and drug delivery. He received his Ph.D. in bioengineering from MIT, and MASc (2001) and BSc (1999) in chemical and biomedical engineering from University of Toronto.
http://www.tissueeng.net/lab/
Ali Khademhosseini is an Assistant Professor of Medicine and Health Sciences and Technology at Harvard-MIT's Division of Health Sciences and Technology and the Harvard Medical School. His research is based on developing micro- and nanoscale technologies to control cellular behavior with particular emphasis in developing microscale biomaterials and engineering systems for tissue engineering and drug delivery. He has published over 60 peer reviewedpapers, 75 abstracts and 14 issued or pending patents. He has received multiple awards including the Coulter Foundation Early Career (2006), Outstanding research mentor at MIT (2004), outstanding researcher in polymer science by OMNOVA / MIT (2005) and outstanding graduate student by Biomedical Engineering Society (2005). He received his Ph.D. in bioengineering from MIT (2005), and MASc (2001) and BASc (1999) degrees from University of Toronto both in chemical engineering.
Source:
Ali Khademhosseini, MASc, PhD, of the Harvard-MIT Biomedical Engineering Center at Brigham and Women’s Hospital, has been recognized by Technology Review magazine as one of the world’s top innovators under the age of 35. Khademhosseini was honored for his pioneering work in the development of novel tissue engineering and cell culture approaches. Selected from more than 300 nominees by a panel of expert judges and the editorial staff of Technology Review, the TR35 is an elite group of accomplished young innovators who exemplify the spirit of innovation in business, technology and the arts.
Source:
Ali Khademhosseini, PhD, of BWH’s Center for Biomedical Engineering within the Department of Medicine, and students from The Harvard-MIT Division of Health, Sciences and Technology have developed a strategy for generating cross-linked hydrogel microstructures – defined as water-soluble, natural polymers – that can be controlled in shape and size for use in drug delivery and tissue engineering. These findings appear online and in the November 29, 2006 print issue of the Journal of the American Chemical Society.
Hydrogels are widely used in biomedical applications because they are flexible in function as a result of their significant water content. Until now, however, it has been difficult to make these variable microstructures from many types of hydrogels that crosslink by a chemical linker. Dr. Khademhosseini and his lab were able to create cross-linked hydrogel microstructures that can be controlled in shape and size by using a micromolding approach combined with the controlled release of the chelating agent.
By having the ability to manipulate cross-linked hydrogel microstructures, researchers can control how particles interact with the body (in drug delivery) and group together to create structures (in tissue engineering). Next steps for the researchers include using these microstructures to generate tissues.
The Coulter Foundation, the Center for Integration of Medicine and Innovation and Technology (CIMIT), and the Charles Stark Draper Laboratory funded this research.
Source:
*Cultivation of human embryonic stem cells without the embryoid body step enhances osteogenesis in vitro.
*Fabrication of Multiphenotye cell arrays within reversibly sealed microfluidic channels for highthroughput analysis.
*“Fabrication of nanostructures of poly(ethylene glycol) and its application to protein and cell patterning”.
*Microencapsulation of cells within shape-controlled microgels as building blocks for tissueengineered organs.
*Microengineering the murine embryonic stem cell environment.
*Multi-phenotype cell arrays inside microfluidic channels.
*Odontogenic fate of stem cells
printer-friendly"
Author(s):
Jianjun Cheng, Christopher H. Yim, Benjamin A. Teply, Dennis Ho , Omid C. Farokhzad, Robert S. Langer
Magnetic responsive particles were designed for use in oral delivery of insulin. Magnetite nanoparticles (12 nm average size) were synthesized and co-encapsulated with insulin into poly(lactide-co-glycolide) (PLGA) microparticles (4.6 2.2 m average particle size) through the double-emulsion method. The spherical structures of resulting microparticles were well maintained at magnetite content 5 wt % or less. Mice were gavaged with 125I-insulin-magnetite-PLGA microparticles and a circumferential trans-abdominal magnetic field was applied forty minutes after administration to retard the transit of the microparticles in the intestinal tract. As control, mice were similarly dosed without the subsequent trans-abdominal magnetic field. Mice were sacrificed, and the intestinal radioactivity was 101% and 145% higher in treated mice versus the control at 6 h and 12 h, respectively. A single administration of 50 unit/kg Humulin R-magnetite-PLGA microparticles to the fasted mice resulted in 66% reduction of blood glucose level in the presence of external magnetic field at 12 h, compared to 27% reduction in the absence of magnetic field.
The Khademhosseini Lab uses a multi-disciplinary approach, to develop microscale and nanoscale technologies with the ultimate goal of generating tissue-engineered organs and controlling cell behavior.
Education of Professor Khademhosseini September 2001- Massachusetts Institute of Technology (MIT) April 2005 Ph.D. in Bioengineering PhD Thesis: “Nanoscale and microscale approaches for engineering the in vitro cellular microenvironment”; Supervisor: Robert Langer |
• Cumulative GPA: 5.0/5.0 September 1999- University of Toronto (U of T) August 2001 M.A.Sc. in Chemical Engineering and Applied Chemistry in Collaboration with Institute of Biomaterials and Biomedical Engineering (IBBME) |
• Masters Thesis: “In vitro study of bone marrow derived progenitor cells in liverlike microenvironments” ; Supervisor: Peter Zandstra and Michael Sefton September 1995- University of Toronto May 1999 B.A.Sc. in Chemical Engineering and Applied Chemistry and Collaborative Program in Environmental Engineering (graduated with honors) • Undergraduate Thesis: “A novel method to conformally coat mammalian cells using magnetically driven beads”; Supervisor: Michael Sefton • Cumulative Average, 1996-99: 88% (4th year average: 93% - rank: 2 of 73) |
FUNDING: Active: 2007-2009 National Institute of Health: R21 Microengineering the murine embryonic stem cell environment; Role: PI 2007-2010 ISN Microencapsulation of cells within shape-controlled microgels as building blocks for tissue engineered organs; Role: co-PI 2006-2007 CIMIT Microencapsulation of cells within shape-controlled microgels as building blocks for tissue engineered organs; Role: PI 2006-2008 Coulter Foundation Microscale bottom-up cardiac tissue engineering; Role: PI 2006-2007 Draper Laboratory Multi-phenotype cell arrays inside microfluidic channels; Role: co-I 2006 National Institute of Health: P20 Systems approach to bioengineering of the tooth; Role: co-PI 2005-2006 Draper Laboratory High-Throughput Multi-Phenotype Cellular Arrays on Surfaces or within Microchannels for Bioanalytical and Diagnostic Devices and Tissue Engineering; Role: co-I 2006-2008 Harvard Stem Cell Institute Odontogenic fate of stem cells; Role: co-PI |
Fujifilm Honeycomb Film : DigInfo
Bone Marrow Transplant - Saving Subash
Seoul National University
Professor Peter W. Zandstra:
Institute of Biomaterials and Biomedical Engineering
Department of Chemical Engineering and Applied Chemistry
University of Toronto
Stem cells have generated much excitement as a potential source of cells for cell-based therapeutics because of their ability to self-renew (divide while maintaining stem cell properties) and differentiate into functional cells (such as blood or heart cells). Consequently, stem cells may serve as a renewable source of cells for regenerative medicine. Despite this enormous potential, significant challenges remain in order to translate the demonstrated biological properties of stem cells into robust and efficacious therapies. We hypothesize that a quantitative systems-based approach, integrating both cell population dynamics and signaling networks involved in individual cell fate decisions, is required to accurately predict and control the generation of cells and tissues from stem cells. This presentation will review some of our efforts to guide the design of novel technologies for stem cell-based therapies, as well as discuss common themes and future challenges in the regulation of cell fate in multicellular systems.
Michael J. Embryo
CIRCULATION
Names of the following videos (left to right) are:
1)Stem Cell Research: Beyond Hype,
Real Hope
2)Stem Cell Research Documentary
3)Stem Cell Patient Interview
4)David Prentice on Amniotic Stem
Cell Research
5)Break-thru on Stem Cells with
StemEnhance
6)The StemTech Story
7) Zebrafish Research
8)How to Make Stem Cells
9)Stem Cell Research
10)Stem Cells - Once and for all
Understanding Embryonic
Stem Cells Part 1 of 6
John D. Gearhart, Ph.D. for funding
embryonic research
Names of the following videos (left to right) are:
1)Stem Cells and Human Cloning
2)Lifting the Ban: The Struggle
for Stem Cells (II)
3)Lifting the Ban: The Struggle
for Stem Cells (I)
4)Cell Sense
5)Embryo development video
6)Development of egg cells
7)ICSI
8)embriologia de mamiferos paulo
9)Embriologia cardiaca
10)Intracytoplasmic Sperm Injection
(ICSI) - Atlanta Georgia